Aluminium Iodide
Aluminium iodide is a chemical compound that has the composition of aluminium and iodine. Aluminium iodide is one of the strong lewis acid and it acts as a absorbent in the atmosphere. The hexahydrate is another form of the aluminium iodide that is obtained by the reaction of metallic aluminium or aluminium hydroxide with hydrogen iodide or hydroiodic acid. The systematic IUPAC name is known as aluminium iodide. The chemical or molecular formula of aluminium iodide is AlI3. The other name is aluminium triiodide.
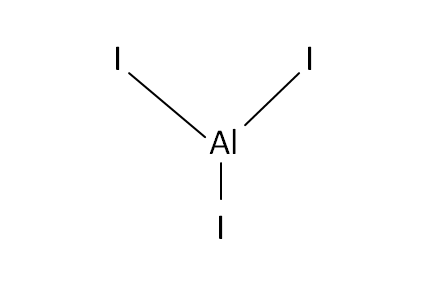
Structural Formula
This is the structural formula of the aluminium iodide:
Chemical Formula
The chemical formula of the aluminium iodide is AlI3.
Preparation Method
The aluminium iodide is prepared by the triiodide is heated at the unstable temperature that gives the formation of aluminium iodide and diiodide as the byproduct. It is mainly used in the particular reagent for the certain kinds of bonds such as C-O and N-O bonds. When it deoxygenates epoxides and it cleaves the aryl ethers.
3 AlI → AlI3 + 2 Al
Physical Properties
| Melting point | 188.28C |
| Boiling point | 382C |
| Molecular weight | 407.695g/mol |
| Density | 3.98g/cm3 |
| Solubility in water | Soluble |
| Crystal structure | Monoclinic |
| Appearance | White or yellow powder |
Chemical Properties
Aluminium iodide is bitter in taste. It looks like a white or yellow powder in the appearance. The forms of monomeric and dimeric is characterized at the gas phase. The melting and boiling point has been very low. It has a low density. It is partially soluble in hydrolysis.
Uses
Aluminium iodide is a catalyst used in certain organic reactions. It is also used in aerosol when treating animal stalls. It is used for animals for treatment. Aluminium iodide is used in organic compounds to make the chemicals. It is used as an additive for electrolytes in the lithium-ion batteries.