Ammonium Phosphate Formula
The ammonium phosphate is a salt that can be obtained from reacting with ammonia and phosphorous acid. It is a colourless and in the form of crystalline solid. It is unstable in nature. It has consists of distinct physical and chemical properties which is known as basic and that is differentiate from the chemical compounds and salts. The systematic IUPAC name is known as ammonium phosphate. The chemical or molecular formula of ammonium phosphate is (NH4)3PO4. The other names is also known as triammonium phosphate.
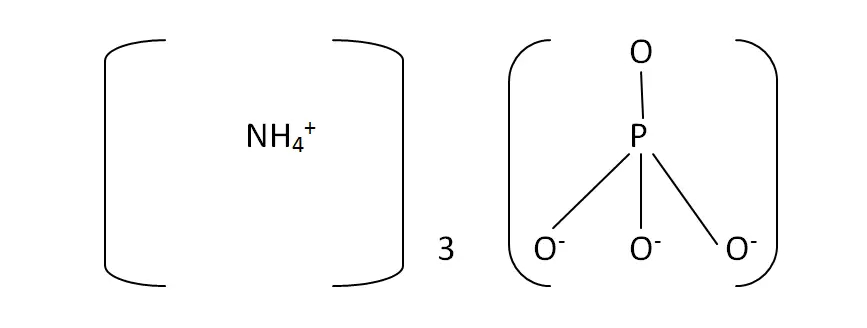
Structural Formula
This is the structural formula of the ammonium phosphate:
Chemical Formula
The chemical formula of the ammonium phosphate is (NH4)3PO4.
Preparation method
When the ammonia is reacts with the phosphoric acid it gives a result called ammonium phosphate. When it is boiled in water, the solution will losses the ammonia which is evaporated as the gas. The ph ranges of the ammonium phosphate is between 4-4.5. then this salt is converted to the diammonium hydrogen phosphate.
H3PO4 + 3NH3 → (NH4)3PO4
Physical Properties
| Molecular weight | 149.09g/mol |
| Solubility in water | 58.0g/100mL(25C) |
| Appearance | White tetrahedral crystals |
| Solubility | Insoluble in acetone |
Chemical Properties
It has anhydrous in nature. It is combined with the nature of the compounds due to highly unstable. These can be divided into three categories dependent on the chemicals compound. They are phosphoric acid and super phosphoric acid, normal superphosphate and triple superphosphate, ammonium phosphate in granular form. It has highly insoluble in acetone chemicals. These are the hazardous substances and used carefully.
Uses
Ammonium phosphate is used to manufacturing for fertilizers that is given to plants for their growth. It can be used as a plant revitalize. Many organic and inorganic compounds used the ammonium phosphate as a solvent for the explosive process. It is also used to the ingredient for chemical extinguisher at the emergency situation. it has increased the nutrient for the any type of soils at regular interval of time.