Strontium Sulfate
Strontium sulfate is an organic compound that has consists the strontium and sulfate. It is more soluble in the nitric acid, dilute HCl and alkali chloride. It naturally occurs in the environment. These strontium sulfate is used for the manufacturing the salts like the other chemical compounds. The systematic IUPAC name is known as strontium sulfate. The chemical or molecular formula of strontium sulfate is SrSO4. It is also known as Celestine.
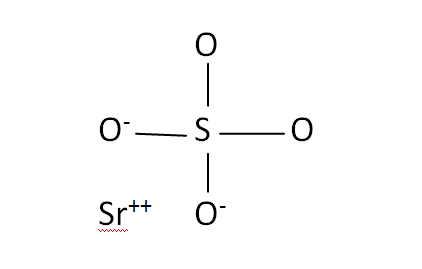
Structural Formula
This is the structural formula of the strontium sulfate:
Chemical Formula
The chemical formula of the strontium sulfate is SrSO4. It has consists of one strontium and four sulfate ions.
How it is Prepared
In the laboratory we can add the dilute sulphuric acid with the strontium oxide it produces the strontium sulfate is the product and water is the byproduct. It appears as the white orthorhombic crystals. The chemical reaction is given as follows. They are
SrO + H2SO4 → SrSO4 + H2O
Physical Properties
| Melting point | 1606C |
| Boiling point | Decomposes |
| Molecular weight | 183.68g/mol |
| Density | 3.96g/cm3 |
| Solubility in water | 0.0135g/100mL |
| Refractive index | 1.622 |
| Crystal structure | Orthorhombic |
| Magnetic susceptibility | -57.9×10-6cm3/mol |
| Appearance | White orthorhombic crystals |
| Solubility | Slightly soluble in acids. Insoluble in alkalis and ethanol. |
Chemical Properties
Strontium sulfate is a non inflammable substances. it is hazardous to the environment. Because it has high melting point. The magnetic susceptibility of strontium sulfate is very low. It is insoluble in alkalis and ethanol. It looks like a orthorhombic in the structure. it has low density and high molecular mass.
Applications
Strontium sulfate is used to manufacturing the ceramics and glasses. It is mostly used for refining the zinc metals. It is mainly used for flares purposes. Strontium sulfate can making the white artist pigment.