Sodium Nitrate
Sodium nitrate is an inorganic sodium salt that has consists of one nitrogen bonds with the three oxygen bonds. While the sodium acts as the cations and the nitrate acts as an anions. When the single bonded atoms is reacted with the oxygen atoms it decomposes over the heat temperature. At that time the electronics are displaced from the nitrite ion to the sodium nitrate. It is the compostion of the sodium and nitrate. The systematic IUPAC name is known as sodium nitrate . The chemical or molecular formula of sodium nitrate is NaNo3. The other names are peru saltpeter, soda niter and cubic niter.
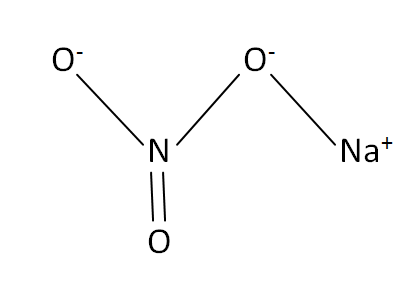
Structural Formula
This is the structural formula of the sodium nitrate:
Chemical Formula
The chemical formula of the sodium nitrate is NaNo3.
Preparation Method
Sodium nitrate can be prepared by the process of exothermic reactions. When the nitric acid is reacted with the sodium carbonate or sodium bicarbonate it gives the formation of sodium nitrate. Carbon dioxide and water as the byproduct. By mixing the stoichiometric compounds it further reacts with the sodium hydroxide with the small amount of ammonium nitrate.
2 HNO3 + Na2CO3 → 2 NaNO3 + H2O + CO2
HNO3 + NaHCO3 → NaNO3 + H2O + CO2
Physical Properties
| Melting point | 308C |
| Boiling point | 380C |
| Molecular weight | 84.99g/mol |
| Density | 2.257g/cm3 |
| Solubility in water | 73g/100g water (0C) |
| Refractive index | 1.587 |
| Crystal structure | Trigonal and rhombohedral |
| Magnetic susceptibility | -25.6×10-6cm3/mol |
| Appearance | White powder or colourless crystals |
| Odor | Sweet |
| Viscosity | 2.85cP |
Chemical Properties
Sodium nitrate is the non combustible and very highly soluble substances. It has bitter in taste. It slightly dissolves in pyridine and insoluble in acetone. It has colourless and odourless compounds. It is trigonal and rhombohedral in structure.
Uses
Sodium nitrate is a food additive and also acts as a food preservative. NaNo3 is a component used for storage and transfer the heat to solar power plants. It is also used as a oxidizer in fire works. The fertilizers are manufactured for the plant growth.