Sodium Formate
Sodium formate is a white crystalline powder that is used as a reagent in the organic chemistry. It has the combination of the sodium and formate. The sodium is positively charged and the formate is the negatively charged. It acts as a proton acceptor in the synthesis of pharmaceuticals. It manufacturing the other chemicals as the intermediate between the reactions. Sodium formate has a variety uses and to improve the flavor and texture of food. The systematic IUPAC name is known as sodium formate. The chemical or molecular formula of sodium formate is HCOONa.
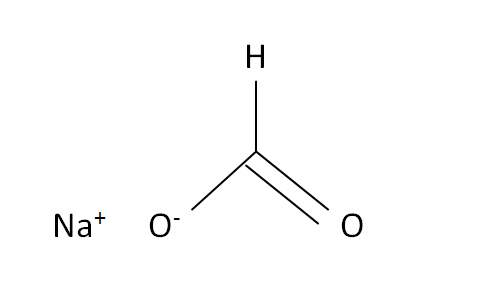
Structural Formula
This is the structural formula of the sodium formate:
Chemical Formula
The chemical formula of the sodium formate is HCOONa. It has consists of one sodium, one hydrogen and two oxygen bonds.
Preparation Method
Sodium formate can be prepared by the reaction between formic acid and sodium carbonate. It obtained the result of sodium formate as the product and sodium chloride and water as the byproduct. By fractional crystallization it could be preferred for the low aqueous solubility and separately by the preparation of chloroform. The chemical reaction is given as follows.
CHCl3 + 4NaOH → HCOONa + 3NaCl + 2H2O
Physical Properties
| Melting point | 253C |
| Boiling point | Decomposes |
| Molecular weight | 68.007g/mol |
| Density | 1.92g/cm3 |
| Solubility in water | 43.82g/100mL |
| Refractive index | 1.456 |
| Appearance | White granules deliquescent |
| Solubility | Insoluble in ether and soluble in glycerol, alcohol and formic acid. |
Chemical Properties
Sodium formate is a hazardous chemical compound and that has been found in the many industrial and consumer products. It cause to irritation of the skin, respiratory problems and cancer diseases. It looks like a white granules deliquescent. It is insoluble in ether and soluble in glycerol, alcohol and formic acid.
Uses
Sodium formate is a buffering agent for strong mineral acids. It also a food additive agent. sodium formate is used as cryoprotectant for x ray diffraction. It could reduce the effects of radiation damage.