What Is Semiconductor | Intrinsic Semiconductor | Extrinsic Semiconductor
Materials
When we talk about materials in electronics we compare them by using current carrying capacity and they can be classified into three types. They are
- Conductors
- Semiconductors
- Insulators
Conductors
Conductors are the materials having excellent current carrying capacity and good flow of current such as copper wire.
Semiconductors
Semiconductors as the name suggest partial or semi conduction of the current or its current carrying capacity between conductor and insulator such as germanium.
Are you looking for PCB manufacturing companies near mountain view?. Then APCT.com is the best Printed circuit board company which have over 340 employees, 4 manufacturing sites, and over 150,000 square feet of combined factory space.
Insulators
Insulators are the materials of bad conductors of current and do not allow follow of current to them as they do not have current carrying capacity such as wood.
History of SemiCondutor:
The term semiconductor was first used by Alessandro Volta (1782). The first documented observation of semiconductor effect in that of Michael faradays in 1833 noticed the silver sulphide decreased with temperature. Which was different then the temperature observed in metal.
Intrinsic Semiconductor | Extrinsic Semiconductor
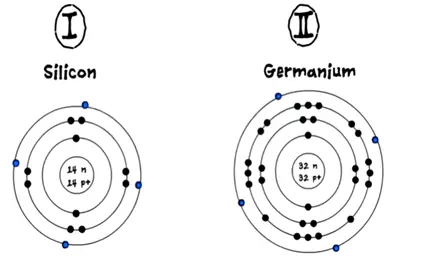
Semiconductors are divided into intrinsic and extrinsic semiconductor. ISC means purest form. These types of semiconductors are in the purest form by the nature of their existence. The common semiconductor is used silicon and germanium. They have four valance electrons orbiting in its outermost shell.
We can further understand the electrons by structure of its atom. However atom needs eight electrons in outer shell to become stable.
Let’s see how semiconductor atom acquires additional four electrons and became stable.
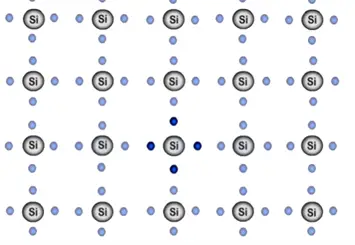
First look at this process with help of one atom. This atom already has its four valance electrons and requires additional four electrons to become stable. Which it shares neighboring atom so that every silicon atom has eight electrons in its outermost shell.
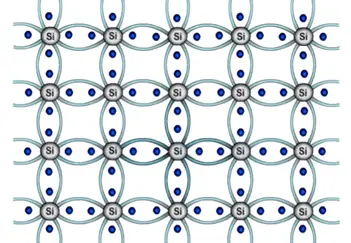
The millions of silicon atoms bonded together and they form semiconductor structure which look like below figure.
All these atoms setup a bond with each other which is called covalent bond. These bonds are so strong that electrons fail to break the band at zero kelvin but as the temperature increases the electrons absorbs the heat energy and able to break the bond once the bond is broken electron become free to carry the current.
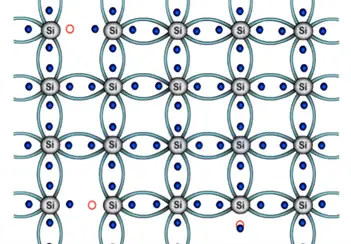
We can get defency of the electron in the structure hence there is an empty space formed which can also refer to as hole. Therefore we consider holes to be positively charged. Electrons here are negative empty hole is positive.
The immediate neighboring electron attracted towards the hole and fills its place. Thus equating empty space or hole the previous position the process will continue in the entire structure in a random manner.
Intrinsic Semiconductor:
How are they different from the intrinsic semiconductor?
Intrinsic semiconductor is a pure form of semiconductor.
Extrinsic semiconductor is impurities need to be added to improve conductivity of these semiconductors.
Extrinsic semiconductors are divided into two types, they are
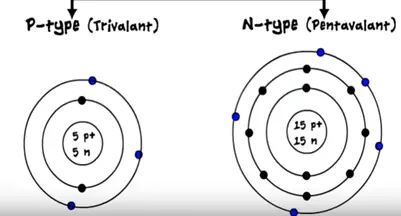
- P-type ( trivalent)
- N-type ( pentavalent)
The three electrons operating in the outermost shell of the atom is called trivalent. On the other atom it has five atoms in the outermost shell of the atom so it is pentavalent.
The common example of the trivalent is the boron, calcium and indium and pentavalent are phosphorous arsenic.
The process of adding impurities to intrinsic semiconductor is called doping. The impurities improves the conductivity of these semiconductors are called as doping.
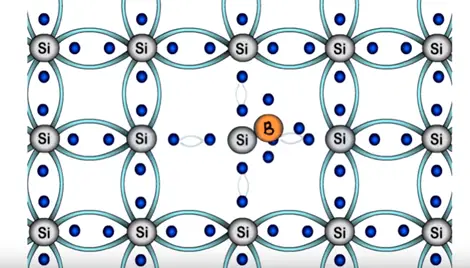
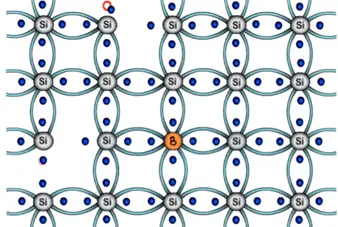
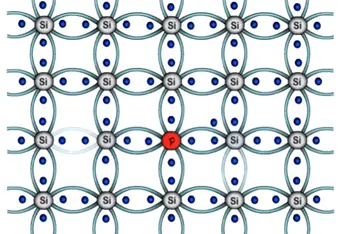
In case of trivalent three electron of boron will form three bonds with silicon and fourth bond of silicon? There will be one empty space or hole. And seen previously immediate neighboring electron will attracted towards the hole thus creating another empty space in its previous location.
Simultaneously the electrons of the silicon atom attain thermal energy keep breaking covalent bonds at room temperature. Thus creating further free electrons this movement will go on same manner. Therefore number of holes generated in this structure dominates over the electrons. Hence structure is called as P-type semiconductor.
Extrinsic Semiconductor:
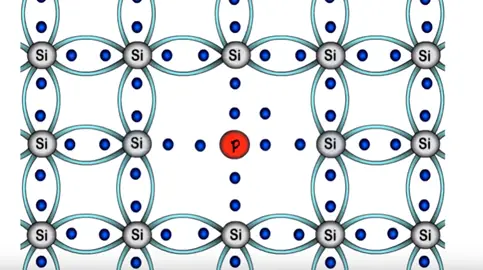
It is a N-type pentavalent semiconductor. In the atomic structure of pentavalent semiconductor will have five electrons in outermost shell silicon requires only addition four electrons to attain stability.
In pentavalent atom we have one extra electron as valance electron. We get a negative charge due to the extra electron hence its name in N-type semiconductor.
Common example of N-type pentavalent impurities phosphor and arsenic
When we add pentavalent impurities to semiconductor the phosphorous atom forms four bonds with silicon atoms to filling the needs of silicon.
There is one electron remains that form any bond and remains free the free electrons readily available for conduction. This electrons rotates randomly around phosphorus atom following an circular motion.